 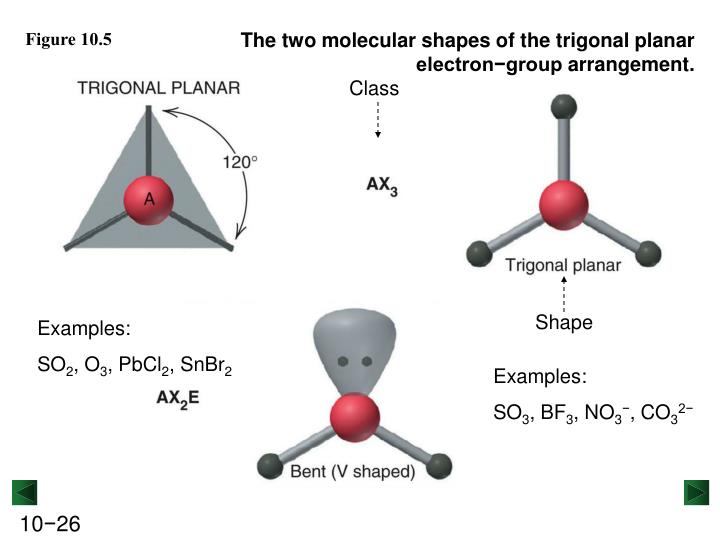
SeO 2 comprises of one selenium atom and two atoms of Oxygen. SeO2 Molecular Geometry and Shape SeO 2 Valence Electrons.Therefore, the molecules in order of increasing bond angle are (4), (1), (3), (2). And finally, molecule (2), carbon dioxide, is the molecule with the largest bond angles of 180 degrees. So the molecule with the smallest bond angles is molecule (4), chlorine trifluoride, with bond angles of 90 degrees, followed by molecule (1), or water, with bond angles of approximately 104.5 degrees, then molecule (3), sulfur dioxide, with bond angles of 118 degrees. This results in a T-shaped molecule with bond angles of 90 degrees. 
This gives sulfur dioxide a bond angle of approximately 118 degrees.įinally, with molecule (4), chlorine trifluoride or ClF3, we can see from the Lewis structure that the central chlorine has three bonding domains and two lone pairs. Since the central atom in sulfur dioxide has only one lone pair, as opposed to two lone pairs on the central oxygen in water, there is decreased repulsion in sulfur dioxide. However, the bond angle is not 104.5 degrees. Sulfur dioxide is also a bent molecule, as we saw with water. Molecule (3), sulfur dioxide or SO2, has a central sulfur atom with two bonding domains and one lone pair. These bonds minimize the repulsion by maximizing the distance between them, resulting in a bond angle of 180 degrees, making carbon dioxide a linear molecule. Even though the carbon and oxygen atoms are covalently bonded by double bonds, each double bond is still only one bonding domain. The Lewis structure for carbon dioxide shows us that carbon is the central atom of the molecule and has two bonding domains. Let’s look now at molecule (2), or carbon dioxide, which has a chemical formula of CO2. However, because of the additional repulsion from the two lone pairs, the bond angle is decreased slightly to 104.5 degrees, giving the water molecule a bent shape. With four total electron domains around the central oxygen, we might expect angles of 109.5 degrees between each domain, as we would find in a tetrahedron shape. And it contains two bonding domains and two nonbonding domains or lone pairs. The central atom of this structure is oxygen. The Lewis structure for water looks like this. Using this information, let’s start with molecule (1), water or H2O. It is worth noting that a lone pair on a central atom generates more repulsion than a bonding domain, thus resulting in smaller bond angles than expected. A nonbonding domain can also be called a lone pair, which is a pair of valence electrons not shared in a covalent bond. In other words, it is a site where valence electrons are shared between the central atom and an outer atom. A bonding domain is an electron domain on the central atom where bonding occurs. We can predict the three-dimensional shapes by determining the number of bonding and nonbonding electron domains that are situated around the central atom of each molecule. This repulsion is minimized by maximizing the space between electron domains. And this model is used to predict the three-dimensional shape of a polyatomic molecule or ion by assuming the electrostatic repulsion between the pairs of valence electrons will be minimized. This stands for the valence shell electron pair repulsion model. We can use the VSEPR model, or Vesper model, to deduce the molecule shapes. 
To determine the angles of the bonds, we must know the three-dimensional shapes of the molecules. And we must determine the comparative values of the bond angles in order to rank them. This question gives us four different molecules. (1) Water, (2) carbon dioxide, (3) sulfur dioxide, and (4) chlorine trifluoride. 
Put the following molecules in order of increasing bond angle. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
